



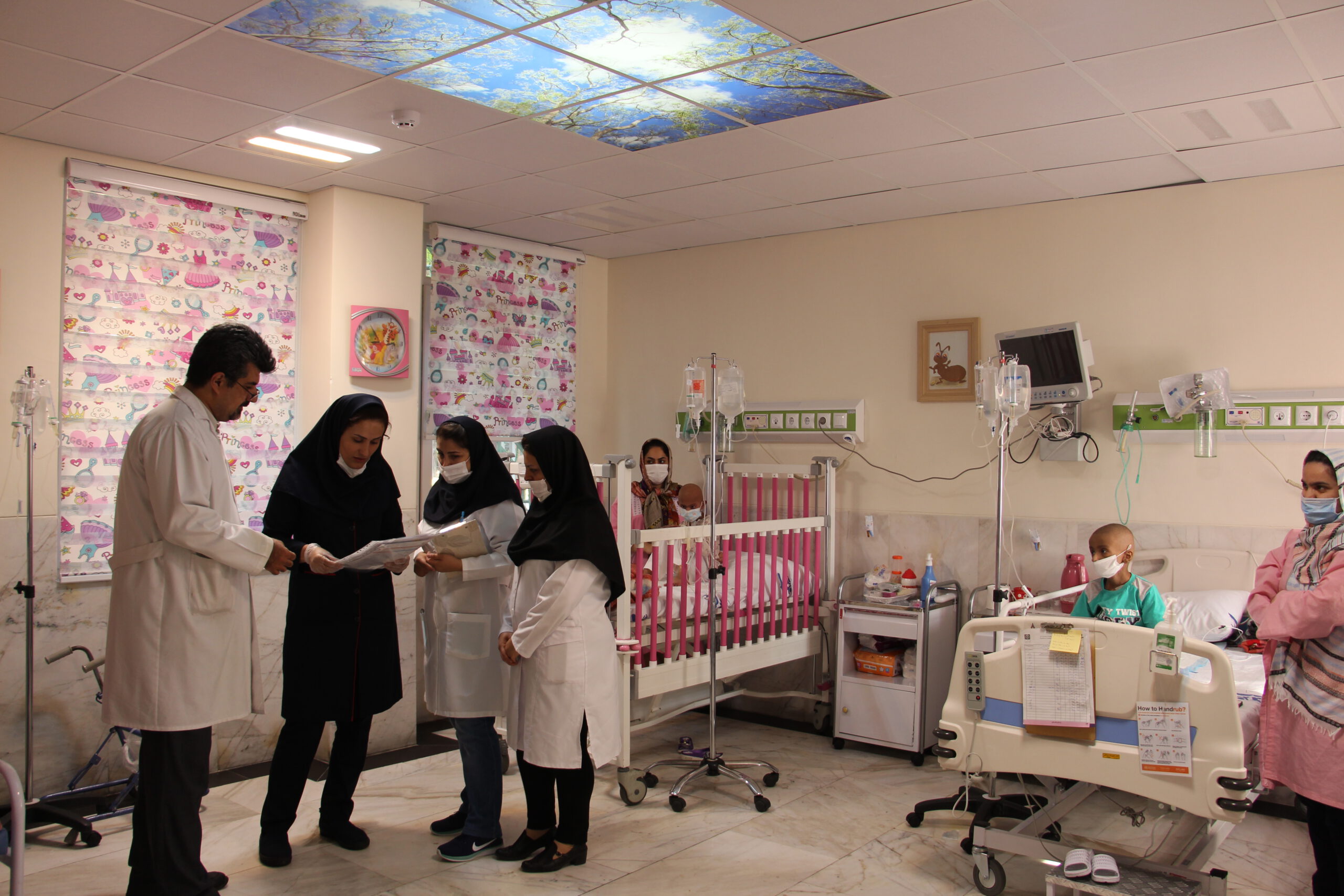
Cell Therapy And Regenerative Medicine And Post Stem Cell Transplantation Ward



Pediatric Cell and Gene Therapy Research Center was established in 2019 to promote research and technology in the field of stem cells and regenerative medicine. The main purpose of this center is to work in the field of gene therapy and cell therapy.
About Us
News
-
NMDP BioTherapies and Cryoport collaborate to Accelerate the Development of Cell and Gene Therapies
NMDP BioTherapies, an organization offering solutions for companies developing and commercializing cell and gene therapies, and Cryoport (NASDAQ: CYRX), a leading global provider of innovative products and services to the fast-growing cell & gene therapy industry enabling the future of medicine for a new era of life sciences, today announced a new strategic partnership. The […]
-
Two Gene Therapy Products for Sickle Cell Anemia Approval by FDA
Two gene therapy products, Casgevy and Lyfgenia, have been approved by the FDA for the treatment of patients 12 years and older with sickle cell anemia. The FDA approved Casgevy to Vertex Pharmaceuticals and approval of Lyfgenia to Bluebird Bio Inc. Casgevy is the first FDA-approved treatment using CRISPR/Cas9 technology, representing an innovative breakthrough in […]
-
Lipid Nanoparticles for Muscle-selective mRNA Delivery
Researchers based at the University of Toronto Leslie Dan Faculty of Pharmacy have developed a novel ionizable lipid nanoparticle (LNP), called iso-A11B5C1, that can deliver mRNA specifically to muscle, while minimizing off-target delivery to other tissues, such as the liver and spleen. Tests in the mouse melanoma model showed that iso-AB11B5C1 LNPs significantly slowed tumor […]
-
Most of Today’s Gene Therapies Rely on Viruses — and that’s a Problem
Around the world, more than 2,000 gene therapies are in development, according to the American Society of Gene and Cell Therapy, which are financially attractive to drug companies. It seems the biggest problem appears to be the viruses ferry genes. Cutting, the geneticist at Johns Hopkins, has high hopes for nonviral delivery vehicles for in-vivo […]
-
Iranian Scientists Made it Possible to Treat Cancer with Gene Therapy : First CAR-T Cell Therapy in Iran
Gene therapy by using CAR-T cells, one of the most advanced methods for treating cancer, has reached the final stages of development thanks to the tireless efforts of professors at Tehran University of Medical Sciences and the researchers of an Iranian research-based start-up company. Currently, only two other multi-national companies offer this treatment worldwide; and […]
-
first transplants of insulin producing cells grown from stem cells
Diabetes researchers have logged another major milestone in their efforts to get people off injected insulin. The first clinical trial in humans reported the “initial success” of transplanting pancreatic cells grown from stem cells to produce insulin. According to the study, 35 per cent of the 17 patients who received implants showed signs of insulin production […]
-
Magnetically guided delivery of therapeutic stem cells into the brain
A minimally invasive method holds promise for the treatment of neurological disorders and injury. Korean researchers have devised a way to remotely direct stem cells to specific areas of the brain to promote neural tissue regeneration. They loaded iron oxide nanoparticles into the cells, which are then guided to the target site by an external […]
-
Interpreting Sameness of Gene Therapy Products under the Orphan Drug Regulations
This guidance provides FDA’s current thinking on determining sameness of human gene therapy products under FDA’s orphan drug regulations for the purpose of orphan-drug designation and orphan-drug exclusivity. This guidance is intended to assist stakeholders, including industry and academic sponsors who seek orphan-drug designation and orphan-drug exclusivity, in the development of gene therapies for rare […]
Contact Us
Address
Children’s Medical Center,
Gharib Ave.
14194, Tehran, Iran






